Last Updated on April 15, 2026
Have you ever wondered how your car starts smoothly every time you turn the key? The secret lies in your car battery, a small but powerful device that generates the energy needed to bring your engine to life.
Understanding how a car battery generates power can give you a clearer picture of what keeps your vehicle running and how to take better care of it. You’ll discover the simple science behind your car battery’s power, why it matters to you, and tips to keep it working longer.
Keep reading—you might be surprised by how fascinating and useful this knowledge can be for your everyday driving.

Credit: ev-lectron.com
Basics Of Car Battery
A car battery stores and provides electrical energy to start the engine. It also powers lights and electronics when the engine is off.
Understanding how a car battery works helps you maintain it and avoid breakdowns.
What Is Inside A Car Battery?
A car battery has several cells filled with an acid solution. These cells create a chemical reaction that produces electricity.
The battery contains lead plates and sulfuric acid. Together, they generate the power needed to start your car.
- Lead plates act as electrodes
- Sulfuric acid acts as an electrolyte
- Chemical reaction produces electric current
How Does A Car Battery Generate Power?
The battery creates power through a chemical reaction between lead plates and acid. This reaction releases electrons, which flow as electricity.
When you turn the key, the battery sends electricity to the starter motor. This starts the engine.
- Lead and acid react to create electrons
- Electrons flow through the battery terminals
- Electricity powers the starter motor
Voltage And Capacity Of A Car Battery
A typical car battery has six cells. Each cell produces about 2 volts, making a total of 12 volts.
The battery capacity shows how much energy it can store. It is measured in ampere-hours (Ah).
| Term | Definition |
|---|---|
| Voltage | Electric force produced (usually 12V for cars) |
| Capacity (Ah) | How long the battery can supply current |
Maintenance Tips For Car Batteries
Keep the battery clean and dry to avoid corrosion. Check the terminals for any dirt or rust.
Make sure the battery is properly charged. Avoid leaving lights or electronics on when the engine is off.
- Clean terminals with a wire brush
- Check battery charge regularly
- Secure the battery to prevent movement
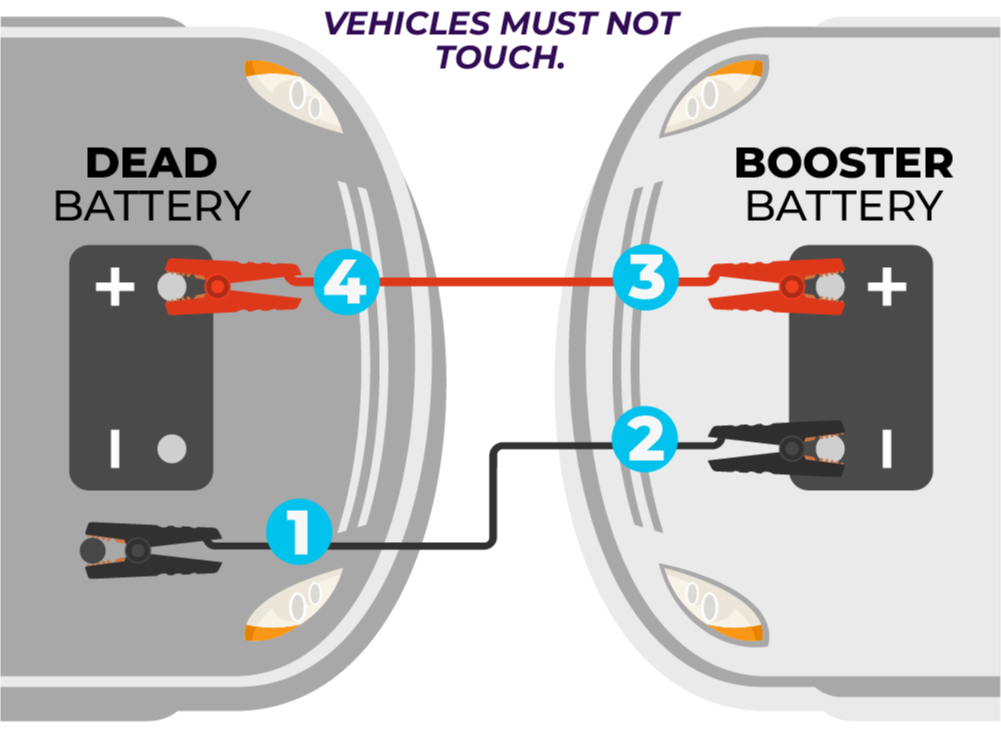
Credit: www.autobatteries.com
Chemical Reactions Inside
A car battery creates power using chemical reactions inside its cells. These reactions produce electrical energy needed to start the engine.
The battery contains lead plates and an acid solution. When the car runs, chemical changes occur that generate electricity.
Basic Components Of A Car Battery
Inside the battery, there are positive and negative plates. These plates are made of lead and lead dioxide. They sit in sulfuric acid.
- Lead dioxide (positive plate)
- Lead (negative plate)
- Sulfuric acid (electrolyte)
How Chemical Reactions Produce Power
When the battery connects to a car, a chemical reaction starts. Lead dioxide and lead react with sulfuric acid. This creates lead sulfate and water.
Electrons move from the negative plate to the positive plate. This flow of electrons creates an electric current.
| Chemical Reaction | Result |
| Lead dioxide + Sulfuric acid | Lead sulfate + Water + Electrons |
| Lead + Sulfuric acid | Lead sulfate + Electrons |
| Electrons flow | Electric current |
Battery Discharge And Recharge
When the car uses the battery power, the chemical reaction creates lead sulfate. This reduces the battery’s charge.
When the car runs, the alternator sends electricity back to the battery. This reverses the chemical reaction. Lead sulfate changes back to lead, lead dioxide, and sulfuric acid.
Role Of Electrolytes
Electrolytes help a car battery create electricity. They are a liquid that lets electric charges move inside the battery.
The battery uses chemical reactions between its parts and the electrolytes to make power. This power starts the car.
What Are Electrolytes Made Of?
Electrolytes in car batteries are usually made of sulfuric acid and water. This mix is called battery acid.
The acid helps carry electric charges between the battery’s plates. The water keeps the acid at the right strength.
How Electrolytes Help Generate Power
Inside the battery, the electrolyte lets ions move freely. Ions are charged particles that carry electricity.
When the battery works, a chemical reaction happens between the plates and the electrolyte. This reaction creates an electric current.
- The electrolyte moves ions from one plate to another
- This movement causes electrons to flow through the car’s circuit
- The flow of electrons is the electric power that starts the car
Why Electrolyte Levels Matter
If the electrolyte level is too low, the battery can’t make enough power. This can stop the car from starting.
Too much water in the electrolyte can also weaken the battery. The right mix keeps the battery strong and working well.
- Check the electrolyte level regularly
- Add distilled water if the level is low
- Avoid spilling battery acid during checks
Safety Tips For Handling Electrolytes
Electrolytes contain acid. This acid can hurt your skin and eyes. Always be careful when handling it.
Wear gloves and goggles. Work in a well-ventilated area. Clean spills right away to stay safe.
Power Flow To The Engine
A car battery stores electrical energy needed to start the engine. It sends power to different parts of the car.
The flow of power from the battery to the engine is important for the car to run smoothly.
Battery Supplies Electrical Energy
The battery creates electrical energy using chemical reactions inside it. This energy moves as electric current.
The battery has two terminals: positive and negative. Electric current flows from the positive terminal.
Starter Motor Uses Battery Power
The starter motor gets power from the battery to turn the engine over. This helps the engine start running.
When you turn the key, the battery sends current to the starter motor through thick cables.
Ignition System Gets Electrical Current
The ignition system uses power from the battery to create a spark. This spark lights the fuel in the engine.
Without this spark, the engine will not run, even if the starter motor turns it.
Alternator Recharges The Battery
Once the engine runs, the alternator makes electricity. It keeps the battery charged during driving.
The alternator also powers other electric parts of the car while the engine is on.
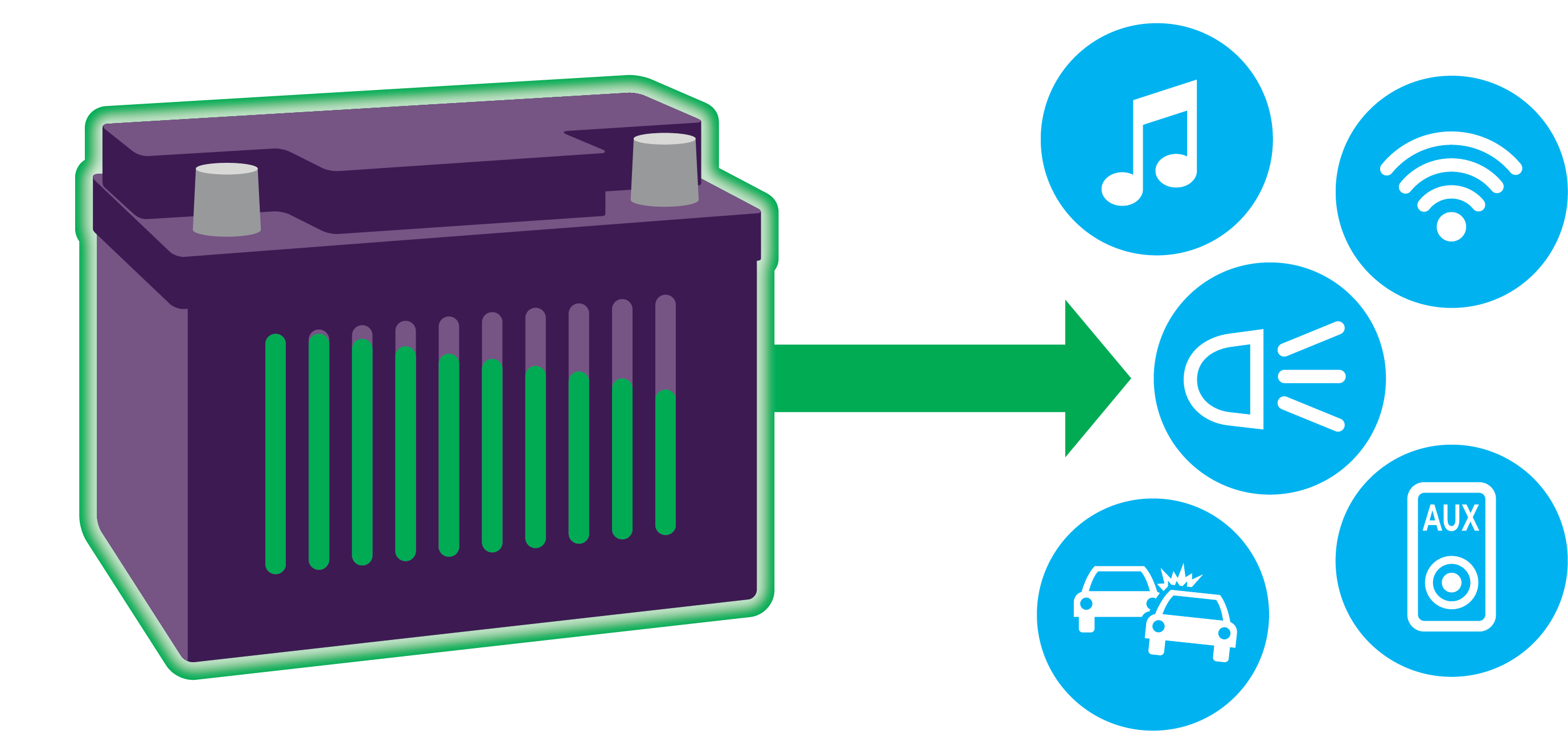
Credit: www.autobatteries.com
Frequently Asked Questions
How Does A Car Battery Create Electrical Power?
A car battery produces power through a chemical reaction between lead plates and acid inside it.
What Role Do Lead Plates Play In Power Generation?
Lead plates act as electrodes that react with acid to release electrons, creating electrical energy.
How Does The Battery Supply Power To The Car?
The battery sends electrical current to the starter and other parts to run the engine and electronics.
Why Is Sulfuric Acid Important In A Car Battery?
Sulfuric acid enables the chemical reactions that produce electrical energy inside the battery.
How Long Can A Car Battery Hold And Provide Power?
A good car battery can store and supply power for several years with proper care.
Conclusion
A car battery works by turning chemical energy into electrical power. This power starts your engine and runs your car’s electronics. Knowing how it works helps you take better care of it. Keep the battery clean and check it often.
A healthy battery means your car stays reliable. Now, you understand the simple but important process behind your car’s power. It’s the small spark that keeps your vehicle moving every day.

